
WebAl2O3 is reduced by electrolysis at low potentials and high currents. If 4.0 × 10^4 amperes of current is passed through molten Al2O3 for 6 hours, what mass of aluminium is. WebElectrolysis and extraction of aluminium Electrolysis involves using electricity to break down electrolytes to form elements. The products of electrolysis can be predicted for a given.
Al2o3 Is Reduced By Electrolysis At Low Potential, `Al_2 O_3 ` is reduced by electrolysis at ow potentials and high currents, If `4.0 xx 10^(4)` ampere, 5.61 MB, 04:05, 801, Doubtnut, 2020-08-16T06:40:43.000000Z, 19, Al2O3, is reduced by electrolysis at low potentialsand high currents, brainly.in, 4160 x 1970, jpeg, electrolysis currents amperes, 20, al2o3-is-reduced-by-electrolysis-at-low-potential, KAMPION
Web`Al_2 O_3 ` is reduced by electrolysis at ow potentials and high currents, If `4.0 xx 10^(4)` amperes of current is passed through molten `Al_2O_3` fro `6` h... WebAl2O3 is reduced by electrolysis at low potentials and high currents. If 4.5 x 104 An of current is passed through molten Al2O3 for 6h, what mass of aluminium is produced?. WebQ. Al2O3 is reduced by electrolysis at low potentials and high currents. If 4.0 × 104 amperes of current is passed through molten Al2O3 for 6 hours, what mass of aluminium. WebAnswer to: Molten Al2O3 is reduced by electrolysis at low potentials and high currents. If 1.5 x 10^4 amperes of current is passed through molten... If 1.5 x 10^4 amperes of current is. WebSecond, cyclic voltammetry is conducted to investigate the reduction behavior of Al 2 O 3, whereby the electrode potential is accurately controlled and. WebCurrent efficiency can be increased and energy consumption can be lowered by low temperature aluminum electrolysis. However, many problems will occur, such as low. WebAl2O3 is reduced by electrolysis at low potentials and high currents. If 4.0 x 104 A of current is passed through molten Al2O3 for 6 h, what mass of aluminium is produced?. WebMolten Al2O3 is reduced by electrolysis at low potentials and high currents. If 1.5 x 10^4 amperes of current is passed through molten Al2O3 for 8.0 hours, what mass. WebAl 2 O 3 is reduced by electrolysis at low potentials and high currents. If 4.0 $$ \times $$ 10 4 amperes of current is passed through molten Al 2 O 3 for 6 hours, what mass of.
Let's see `Al_2 O_3 ` is reduced by electrolysis at ow potentials and high currents, If `4.0 xx 10^(4)` ampere Latest
Currently - `Al_(2)O_(3)` is reduced by electrolysis at low potentials and high current. If `4.0 xx 10^(4)` popular
Read more from Al2o3 Is Reduced By Electrolysis At Low Potential from the discussion earlier
`Al_2 O_3 ` is reduced by electrolysis at ow potentials and high currents, If `4.0 xx 10^(4)` amperes of current is passed through molten `Al_2O_3` fro `6` hours , what mass of alumininum is produced ? (Assume ` 100%` current efficiency gt At mass of ` Al = 27 g ` "mol"^(-) )`.
Must see Al2O3, is reduced by electrolysis at low potentialsand high currents more

Reviews 9. Al2O3 is reduced by electrolysis at lo... - Physical Chemistry Latest

Look Al2O3 is reduced by electrolysis at low potential and high current. If trending

Here Effect of ACD (CR=2.48, 5wt%CaF2, saturated Al2O3 content, 0.6 A/cm 2 trending

Watch Effect of ACD (CR=2.48, 5wt%CaF2, saturated Al2O3 content, 0.6 A/cm 2 Latest

Reviews CVs obtained on W electrode in 1.5KF-AlF 3-Al 2 O 3(sat) melt at trending

Images Al2O3 is reduced by electrolysis at low potentials and high currents going viral

About Water | Free Full-Text | Physico-Chemical, Biological and Therapeutic
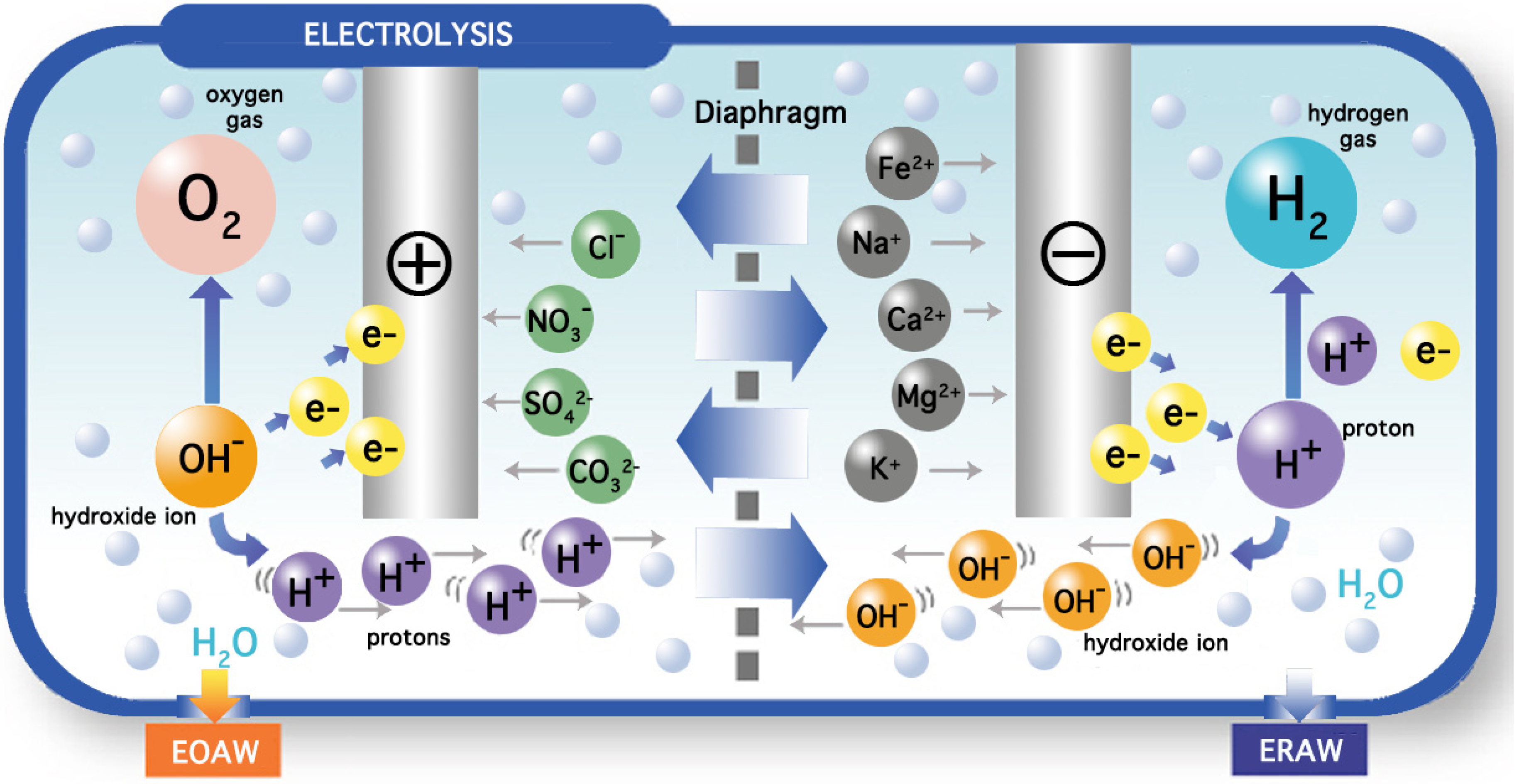
Look (b) Reduced cell-fluid interaction. The results in left column (cell going viral
View Electrochemistry_Notes (Electrolysis+Galvanic +Electronic cell+Faraday popular

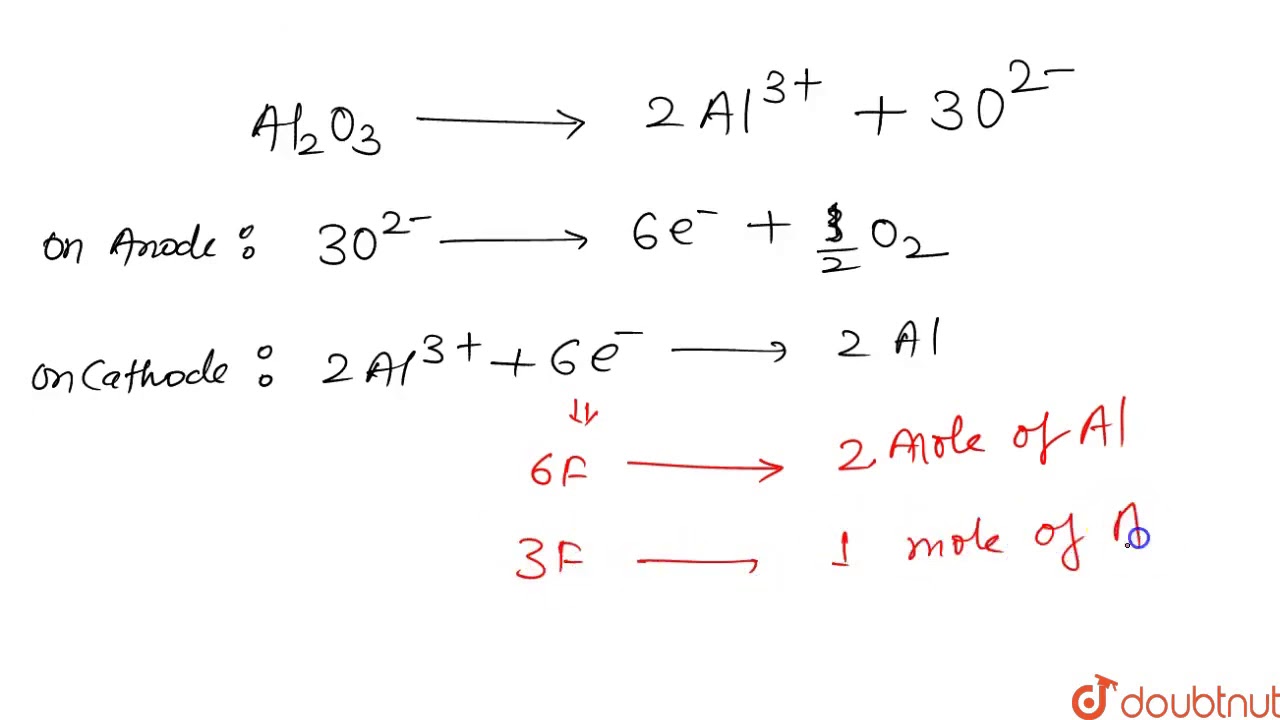
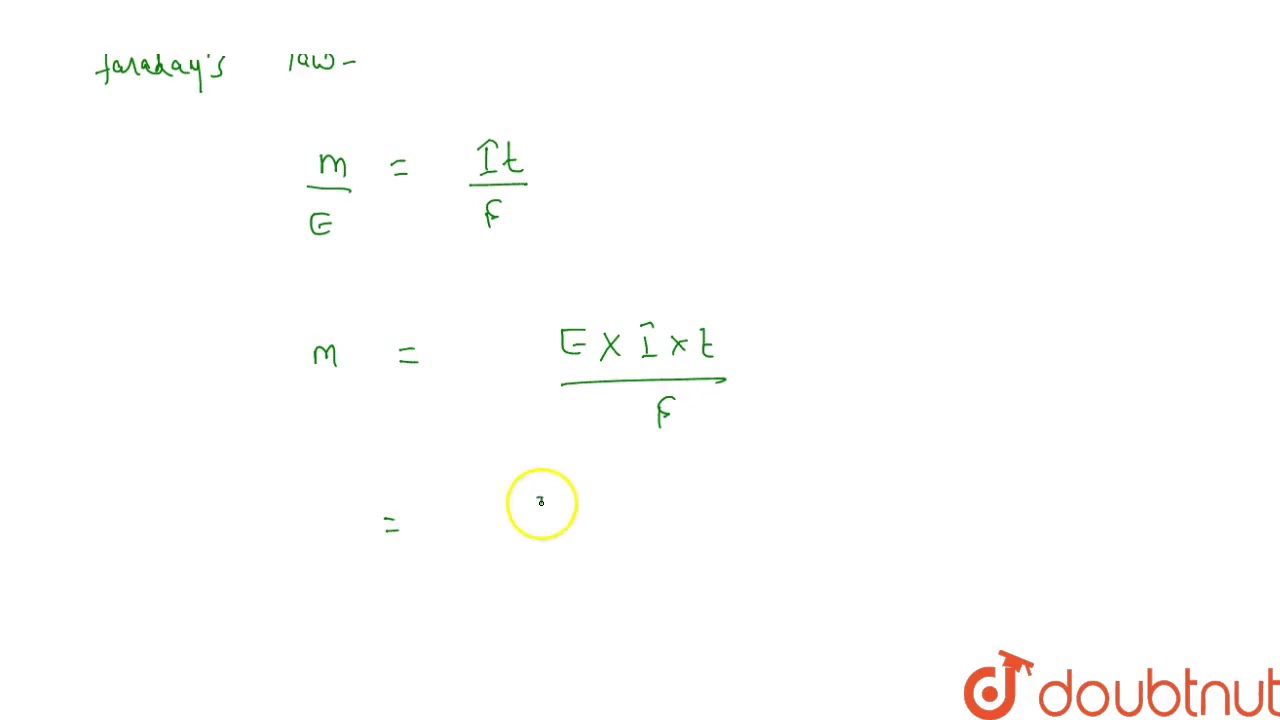
Belum ada tanggapan untuk "Discussion Al2o3 Is Reduced By Electrolysis At Low Potential Popular"
Posting Komentar